Conditions · Cervical cancer, STIs & BV
Gynecologic infection screening on a single sample.
Cervical cancer screening is Qvin's primary clinical menu, with STI panels and bacterial vaginosis on the same molecular platform — all from a single Q-Pad collection event.
The condition
A largely preventable cancer, screened too rarely.
Persistent infection with high-risk human papillomavirus (hrHPV) causes essentially all cervical cancer [1]. Sexually transmitted infections (chlamydia, gonorrhoea, trichomonas, mycoplasma genitalium) and bacterial vaginosis sit alongside HPV as the highest-volume gynecologic infections, often co-occurring. The WHO has set targets for global cervical cancer elimination — 70% of women screened with high-performance assays by age 35 and again at 45 — but most countries are far below the screening coverage needed to hit them [2].
Standard of care
The participation problem.
Standard cervical cancer screening is clinician-collected: a speculum exam with hrHPV testing and/or cytology. The single largest barrier to coverage is participation — women miss appointments, avoid pelvic exams, or live too far from screening services. Self-collection (vaginal swab or first-void urine) is increasingly accepted as a clinically equivalent option, with the WHO and several national programs explicitly endorsing it [2].
Today
With Q-Pad

Why menstrual blood
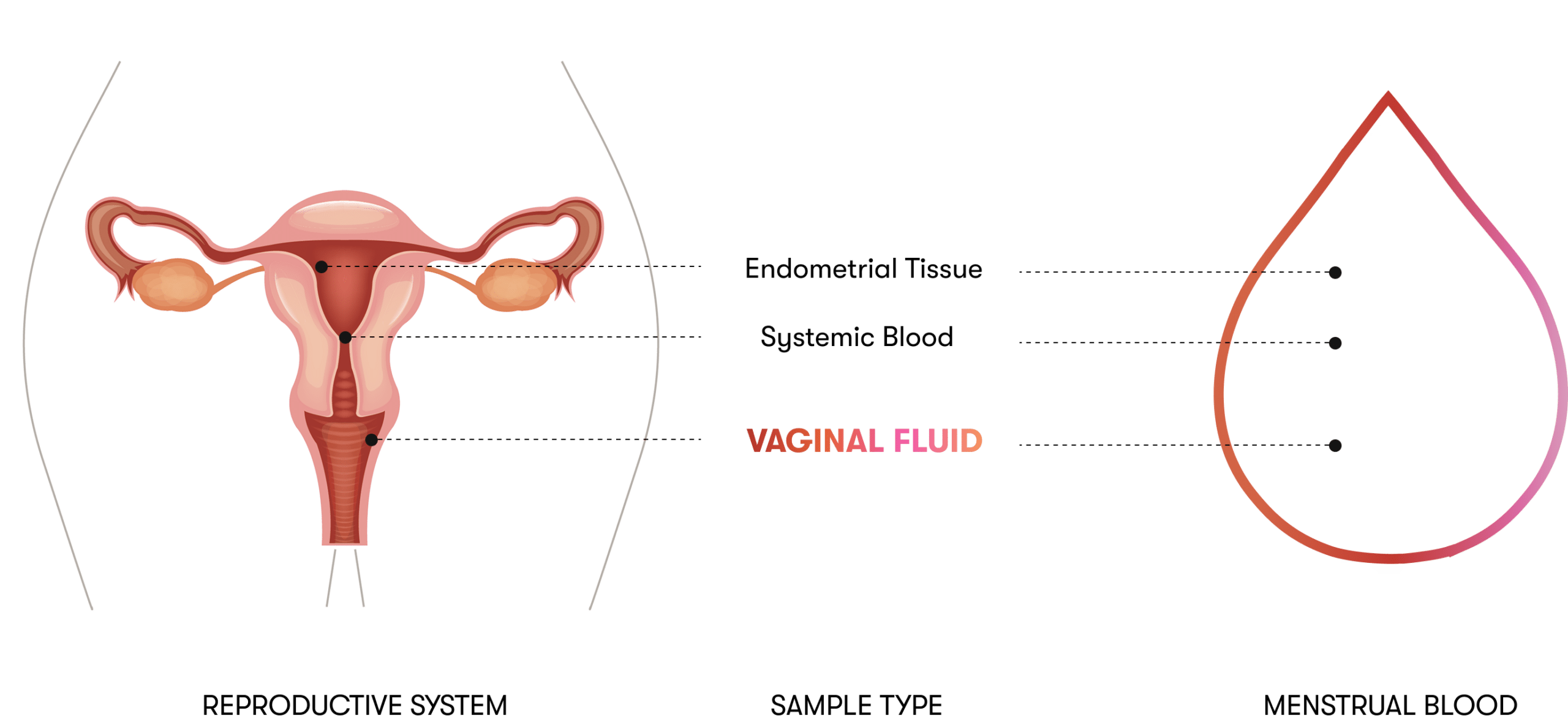
The most concentrated cervicovaginal sample women produce.
Menstrual blood is the most concentrated sample of cervicovaginal biology a woman produces — by definition, it has just transited the cervical canal. It carries cervical and endometrial cells, vaginal microbiota, and any pathogens shedding nucleic acids in that compartment. Validation of HPV genotyping from menstrual blood demonstrates analytical concordance with clinician-collected hrHPV testing on regulator-cleared molecular platforms [3].
Qvin's role
A single sample, three regulator-track menus.
Qvin's lead clinical menu on the Q-Pad is hrHPV genotyping, with STI panels and bacterial vaginosis characterization on the same molecular platform. The clinical case is straightforward: a woman who would otherwise miss a screening visit can collect at home, mail a Q-Pad sample, and receive a clinically actionable result on a regulator-cleared assay. Read more on Collection and Science.
References
Sources
- Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S. Human papillomavirus and cervical cancer. The Lancet 370(9590):890–907, 2007. doi.org/10.1016/S0140-6736(07)61416-0.
- World Health Organization. Global strategy to accelerate the elimination of cervical cancer as a public health problem. WHO, 2020. who.int.
- Naseri S, Rosenberg-Hasson Y, Maecker HT, Avrutsky MI, Park CL. Demonstration of menstrual blood as a viable source for screening and diagnostic clinical applications: a pilot study. BMC Women's Health, 2023. bmcwomenshealth.biomedcentral.com.
Other conditions