Science
Why menstrual blood works.
A composite sample — peripheral blood, endometrial cells and tissue, immune populations, and cervicovaginal fluid — that arrives every cycle by virtue of biology. Dried Menstrual Samples (DMS) inherit the analytical surface of Dried Blood Spots and add gynecological biology on top. The case for the platform rests on this scientific foundation.
Biology
What menstrual blood is.
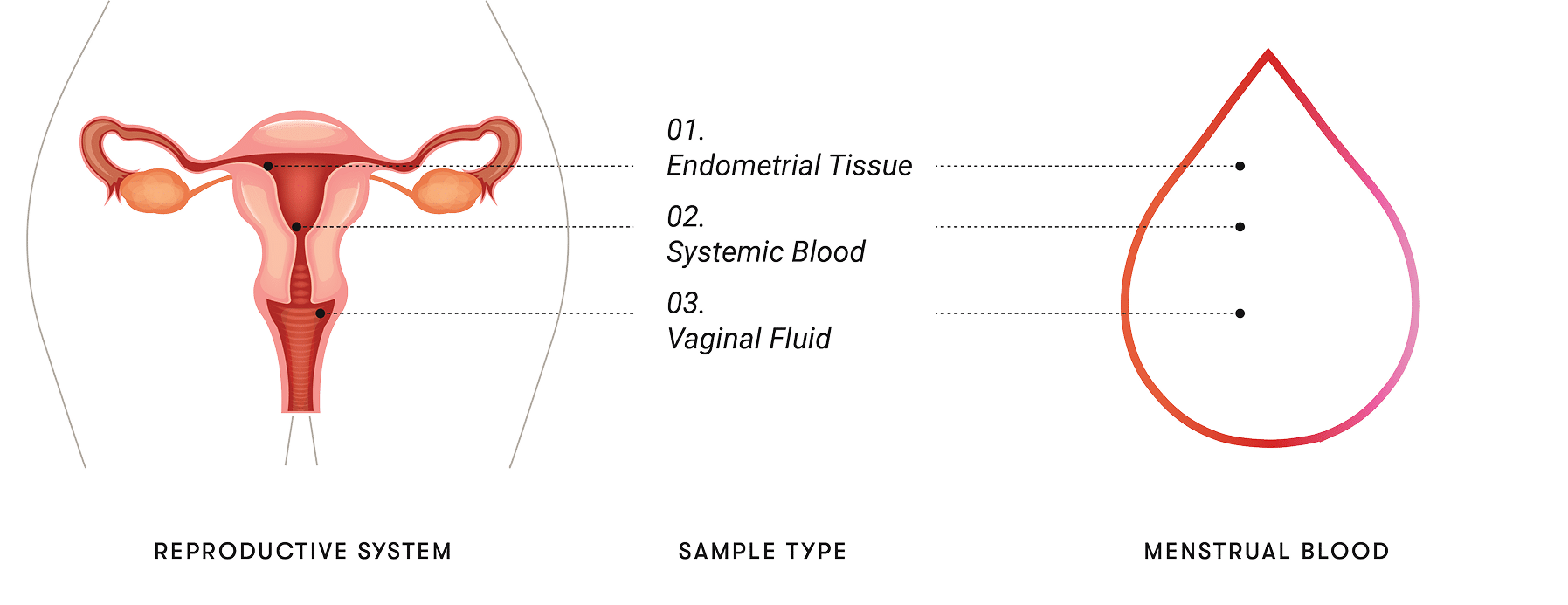
Menstrual effluent is not just blood. It is a biological composite — peripheral blood that has crossed the endometrium, shed endometrial epithelial cells and stromal tissue, immune cells (T cells, NK cells, macrophages), endometrial extracellular matrix, and a contribution of cervicovaginal fluid [1][2]. Proteomic surveys have identified thousands of proteins in menstrual blood, with overlap to and divergence from the plasma proteome [3]; the reproductive-biology literature on endometrial regeneration further details its cellular content [2].
Two properties are clinically distinctive. First, the sample is composite: in the same volume of effluent there is signal from systemic circulation (peripheral blood) and signal from the gynecological tract (endometrium, cervix). Second, the sample is recurring: a substantial subset of women produce it monthly for decades, without intervention, instrumentation, or behavior change. No other clinical sample combines those two properties.
Sample type
Dried Menstrual Samples — and what we know from Dried Blood Spots.
A Dried Menstrual Sample is menstrual blood collected on a substrate and dried at the point of collection. The drying step is what turns a biologically rich but logistically fragile fluid into a stable, mailable, scalable analytical specimen. The sample type is closest in spirit to a Dried Blood Spot (DBS) — a substrate that has supported clinical analysis since Guthrie and Susi's 1963 application to phenylketonuria newborn screening, and which today underpins national newborn screening programs in most high-income countries [4].
Why the DBS comparison matters
Dried Blood Spots have been continuously developed for sixty years. The literature on DBS as an analytical substrate now spans molecular diagnostics (DNA, RNA, methylation), clinical chemistry (HbA1c, lipids, vitamin status), immunology (antibody titers, cytokines), pharmacokinetics (therapeutic drug monitoring), and population epidemiology [5][6]. The infrastructure that enables this — sample handling SOPs, regulatory frameworks, elution chemistries, automated analyzers — transfers directly to DMS. Qvin builds on this prior art rather than reinventing it.
Analytical approaches that work on DMS
On the basis of DBS-grounded methodology and Qvin-led validation, the following analytical surfaces are accessible from a DMS strip:
- Molecular — DNA (PCR, qPCR, NGS), RNA (RT-PCR), and methylation assays. HPV genotyping is the lead clinical assay; STIs, bacterial vaginosis, and broader pathogen panels are downstream [7].
- Chemistry & immunoassay — HbA1c, hormones (TSH, AMH, FSH, LH, estradiol), proteins, lipids. Validated correlations with venous blood at r > 0.9 across multiple targets [8].
- Multi-omics — proteomics, metabolomics, methylomics. Discovery work runs through institutional partners; targets identified are translated into regulator-track assays on the Q-Pad over time.
- Endometrial biology — markers and cell-derived signal that are not accessible from peripheral blood at all. This is the unique surface the Q-Pad opens that DBS cannot.
Validation
What's been shown so far.
Qvin-led and Qvin-collaborator studies have demonstrated that menstrual blood collected on the Q-Pad recapitulates clinically meaningful information from venous draws, with strong concordance for several broad biomarker categories [8]:
- HbA1c — concordance with venous HbA1c suitable for diabetes screening and monitoring.
- Reproductive hormones — TSH, AMH, FSH, LH, estradiol with high correlation across the menstrual cycle.
- HPV genotyping — sensitivity and specificity sufficient for cervical cancer screening, on regulator-cleared molecular platforms.
- Cellular and tissue signal — endometrial markers visible because the menstrual sample contains endometrial material, supporting work in endometriosis, fertility, and gynecologic cancer (early-stage research).
Institutional collaborations
The validation surface expands under collaborations with major research institutions:
- Statens Serum Institut (CYCLOME) — Wellcome Leap–funded program studying the cyclical proteome and methylome of menstrual blood across thousands of participants.
- Stanford University (iPOP) — multi-omic deep phenotyping leveraging menstrual blood as a recurring at-home sample, in the Snyder Lab tradition [9].
- University of Exeter — Wellcome Leap–funded program on menopause biomarkers and the perimenopausal transition.
- Mayo Clinic — multi-program biomarker work spanning gynecologic and systemic health.
Looking forward
A platform that compounds across indications.
A regulator-cleared menstrual blood collection device, paired with an analytical surface anchored in the DBS literature, is a foundation that compounds. Each new validated assay strengthens the case for the next; each regulatory submission builds on the predicate before it; each institutional collaboration adds to the reference base. The science is what makes the rest possible — and the science argues that menstrual blood, properly collected and dried, is one of the most underdeveloped analytical substrates in clinical medicine.
References
Sources
- Bobel C, Winkler IT, Fahs B, Hasson KA, Kissling EA, Roberts T-A, eds. The Palgrave Handbook of Critical Menstruation Studies. Palgrave Macmillan, 2020 — chapters covering menstrual fluid composition. link.springer.com.
- Meng C-X, Andersson KL, Bentin-Ley U, et al. Endometrial regenerative cells: a novel stem cell population. Journal of Translational Medicine 5:57, 2007. doi.org/10.1186/1479-5876-5-57.
- Yang H, Zhou B, Prinz M, Siegel D. Proteomic analysis of menstrual blood. Molecular & Cellular Proteomics 11(10):1024–1035, 2012. doi.org/10.1074/mcp.M112.018390.
- Guthrie R, Susi A. A simple phenylalanine method for detecting phenylketonuria in large populations of newborns. Pediatrics 32:338–343, 1963. publications.aap.org.
- McDade TW, Williams S, Snodgrass JJ. What a drop can do: dried blood spots as a minimally invasive method for integrating biomarkers into population-based research. Demography 44(4):899–925, 2007. doi.org/10.1353/dem.2007.0038.
- Lehmann S, Delaby C, Vialaret J, Ducos J, Hirtz C. Current and future use of "dried blood spot" analyses in clinical chemistry. Clinical Chemistry & Laboratory Medicine 51(10):1897–1909, 2013. doi.org/10.1515/cclm-2013-0228.
- U.S. Food & Drug Administration. 510(k) Premarket Notification database — Q-Pad cleared device record and HPV-related submissions. accessdata.fda.gov.
- Naseri S, Rosenberg-Hasson Y, Maecker HT, Avrutsky MI, Park CL. Demonstration of menstrual blood as a viable source for screening and diagnostic clinical applications: a pilot study. BMC Women's Health, 2023. bmcwomenshealth.biomedcentral.com.
- Chen R, Mias GI, Li-Pook-Than J, et al. Personal omics profiling reveals dynamic molecular and medical phenotypes. Cell 148(6):1293–1307, 2012 — foundational iPOP method paper, Snyder Lab, Stanford. doi.org/10.1016/j.cell.2012.02.009.